 An electron shared by more than two atoms is said to be delocalized. The two π electrons in the nitrate ion are shared by a total of four atoms, one nitrogen atom and three oxygen atoms. If a pair appears in one place in one form, and in a different place in another form, the pair is delocalized. The easiest way to spot delocalized electrons is to compare electron locations in two resonance forms. How do you tell if there are delocalized electrons? So, only option R have delocalized electrons. Since lone pairs and bond pairs present at alternate carbon atoms. In the given options, In option R, electron and bond are present at alternate carbon atoms. Which of the following has delocalized electrons? Delocalized electrons are contained within an orbital that spans several neighbouring atoms. What molecules have delocalized electrons?ĭelocalized electrons are electrons that are not associated with a single atom or covalent bond in a molecule, ion, or solid metal. And that's what we're going to have a look up next localized PI electrons are shared between all atoms in a molecule. Quote from video: The circle in the middle represents delocalized pi electrons. Sp 2 orbital: One of a set of hybrid orbitals produced when one s orbital and two p orbitals are combined mathematically to form three new equivalent orbitals oriented toward the corners of a triangle (hence the designation trigonal planar). With sp 2 hybridization, each carbon atom has an unhybridized atomic p orbital associated with it. Are benzene rings sp2?īecause experimental data shows that the benzene molecule is planar, that all carbon atoms bond to three other atoms, and that all bond angles are 120°, the benzene molecule must possess sp 2 hybridization. Six π- Molecular Orbitals (MO’s) of Benzene. How many degenerate orbitals are in benzene? Notice that the 1s orbital has the highest probability. Below is a diagram that shows the probability of finding an electron around the nucleus of a hydrogen atom. What is orbital structure?ġ) An orbital is a three dimensional description of the most likely location of an electron around an atom. How many delocalized electrons are present?Ĥ π-electrons of double bond and 1 lone pair on N−atom leads to delocalization of π-electrons. The six atomic p orbitals combine to form six molecular π orbitals. There are three antibonding orbitals in benzene. How many antibonding orbitals are in benzene? Both forms contain two delocalized electron pairs. You can see delocalized behavior in resonance forms I and II below. 
Hence, the total number of sp 2 hybrid orbitals is 18. 3 -bonds means that there are 3 sp 2 hybrid orbitals for each carbon. The number of sp 2 hybrid orbitals in molecule of benzene is:īenzene (C 6H 6) has 6 sp 2 hybridized carbons. On average all of the ring bonds are identical. In the diagram below, the p orbitals have combined, and the π-electrons are delocalized. The p orbitals combine side on and the electrons in the p orbitals are described as π-electrons. Consequently, the molecular orbital approach, called molecular orbital theory is a delocalized approach to bonding. What are delocalized molecular orbitals?Īs the name suggests, molecular orbitals are not localized on a single atom but extend over the entire molecule. As shown below, the remaining cyclic array of six p-orbitals ( one on each carbon) overlap to generate six molecular orbitals, three bonding and three antibonding. We know that benzene has a planar hexagonal structure in which all the carbon atoms are sp2 hybridized, and all the carbon-carbon bonds are equal in length. How many delocalised pi electrons are present in benzene molecule ? The delocalised `pi` -molecular orbital in benzene contains three …… What is the orbital structure of benzene? 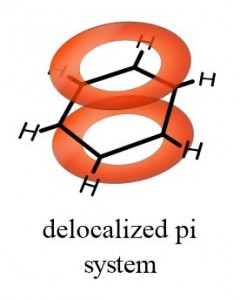
How many delocalized molecular orbitals are there in benzene? What orbitals are delocalized electrons in?ĭelocalization is central feature of molecular orbital theory where rather than the lone pair of electrons contained in localize bonds (as in the valence bond theory), electrons can exist in molecular orbitals that are spread over the entire molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
